Fairfax County has surpassed the halfway mark for COVID-19 vaccinations, as reported cases of the disease caused by the novel coronavirus continue to decline.
According to Virginia Department of Health data, 51% of Fairfax County’s population — or 585,447 residents — have now gotten at least one COVID-19 vaccine shot. That puts the county in line with neighboring jurisdictions in Northern Virginia, including Loudoun (50.4%) and Arlington (51.2%).
After previously trailing by a hair, the county now has now inched past Virginia as a whole in terms of fully vaccinated residents. 35.4% of the county’s population — or 406,383 people — have received all required shots, compared to 35.1% of the state overall.
Virginia has administered at least one dose to more than 4 million people, or 47.1% of its population. 3 million residents have been fully vaccinated.
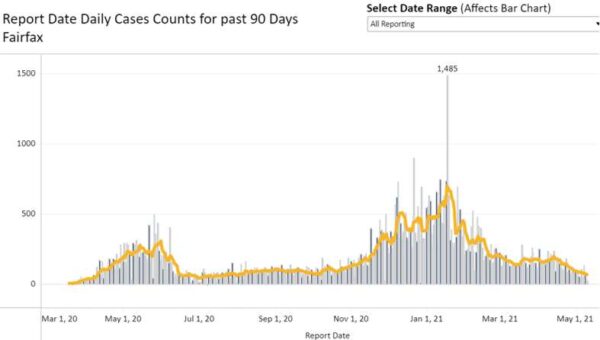
With more people getting vaccinated, Fairfax County’s COVID-19 caseload continues to shrink.
While the number of new cases ticked back up to 126 cases on Friday (May 7), the Fairfax Health District reported just 22 cases today (Monday), the fewest since 21 cases came in on Sept. 28.
The county is now averaging 63.4 cases over the past seven days, bringing the case rate down to a level not seen since Aug. 1, when the weekly average was at 60.6 cases after hovering in the 50s and 60s throughout July.
The Fairfax Health District has now recorded a total of 77,422 COVID-19 cases, 4,053 hospitalizations, and 1,104 deaths.
While demand for the COVID-19 vaccine has started to ebb, Virginia’s push to achieve herd immunity could get a boost if federal officials approve the vaccine for adolescents between the ages of 12 and 15 this week as anticipated.
The Centers for Disease Control and Prevention’s Advisory Committee on Immunization Practices is scheduled to meet on Wednesday (May 12) to discuss recommending that the Pfizer-BioNTech vaccine be approved for 12 to 15-year-olds, according to the Fairfax County Health Department.
Pfizer reported on March 31 that its vaccine has shown a 100% efficacy rate and “robust antibody responses” in a clinical trial with 2,260 participants between 12 and 15 years old, none of whom contracted COVID-19 after getting vaccinated.
The company says it expects to get authorization from the Food and Drug Administration for the expanded use of its vaccine, which is currently approved for people 16 and older, sometime this week.
The Fairfax County Health Department says vaccine will be ready so that parents and guardians can start making appointments as soon as the federal approval comes in.
“The state and local health departments will let everyone know when the vaccine is approved for use among 12-15-year-olds and our appointment scheduling systems will update accordingly,” the FCHD said in a blog post.
Chart via Virginia Department of Health